What is Matter?
Think about everything that you touch or see, every single day. Anything and everything that you can touch or see, is matter. But, what is matter?
The dictionary definition to matter is:
Matter |'matər|
Noun
anything that takes up space and has mass
Anything that takes up space is matter. Even air is matter; even you're made of matter; the whole world is made of matter! The only place where there is no matter, is in a theoretical vacuum. All matter is made up of very small, microscopic particles. You cannot see these particles unless you are looking under a very strong microscope, as they are very small. All particles are bumpy (even on smooth surfaces), and never touch one another. The study of matter, is called chemistry, and scientists who study matter, are called chemists.
The dictionary definition to matter is:
Matter |'matər|
Noun
anything that takes up space and has mass
Anything that takes up space is matter. Even air is matter; even you're made of matter; the whole world is made of matter! The only place where there is no matter, is in a theoretical vacuum. All matter is made up of very small, microscopic particles. You cannot see these particles unless you are looking under a very strong microscope, as they are very small. All particles are bumpy (even on smooth surfaces), and never touch one another. The study of matter, is called chemistry, and scientists who study matter, are called chemists.
The States of Matter
There are three main types of matter: solid, liquid, and gas. The particles in all these states of matter, are the same. E.g. ice (solid), water (liquid) and water vapor (gas), but with a different particle movement, particle arrangement, etc.
The table above is showing us the differences between the three states of matter.
Ability to Compress Break-Down: First, we must understand exactly what compress means, so below is the definition:
Compress /kəmˈpres/
Verb
force in less space
So, compress means to make the same amount of particles, come together, and form into less space. Since the particles in a solid, are already very dense, and do not have much space between them, a solid cannot be compressed much. As liquid is dense, and has some space between the particles, so a liquid can be slightly compressed. As a gas is not dense at all, and the particles are far from one another, and do have space between one another, a gas can be compressed a lot.
Particle Movement Break-Down: The particles in a solid, do not move around much as they do not have much space, as all the particles are close together, due to the particles attracting themselves to one another. The particles in a liquid move around in the area, as they have space within the area, but not enough to go everywhere, as the particles attract other particles to it. The particles in a gas move around all over the place, as they have the space to do so, and the particles are not close enough to be attracted by the other particles.
Shape Break-Down: Since the particles of a solid are packed together, and constantly attracting to one another, these particles cannot move, and thus only stay in one shape (unless of course force is applied, or if it is heated or cooled). The particles of a liquid and gas do not have strong enough attraction, and thus the particles can re-arrange themselves, to take a new shape (not necessarily in a different amount of space, but in a different shape/form).
Volume Break-Down: Since all particles of matter always attract to one another; if the particles of a solid or liquid are constantly close enough to always have the same attraction to another specific particle, then that means that the particles will always be as close together to one another at all times (not necessarily in the same shape), and will always take up the same amount of space, at all times. So, a solid and a liquid, will always have the same volume, at all times. But for a gas, the particles aren't close enough to always have the same attraction to a specific particle, so the particles will not always be as close to one another. So, sometimes the particles will be close together, and take up less space, and sometimes the particles will be far apart, and take more space.
Ability to Flow Break-Down: Since the attraction between particles in a solid is quite strong, this prevents the particles from moving freely, so a solid's particles cannot flow. But, the bonds between particles in a liquid and gas, aren't as strong, so the particles can move freely, and take a new shape (with the same volume of course).
Ability to Compress Break-Down: First, we must understand exactly what compress means, so below is the definition:
Compress /kəmˈpres/
Verb
force in less space
So, compress means to make the same amount of particles, come together, and form into less space. Since the particles in a solid, are already very dense, and do not have much space between them, a solid cannot be compressed much. As liquid is dense, and has some space between the particles, so a liquid can be slightly compressed. As a gas is not dense at all, and the particles are far from one another, and do have space between one another, a gas can be compressed a lot.
Particle Movement Break-Down: The particles in a solid, do not move around much as they do not have much space, as all the particles are close together, due to the particles attracting themselves to one another. The particles in a liquid move around in the area, as they have space within the area, but not enough to go everywhere, as the particles attract other particles to it. The particles in a gas move around all over the place, as they have the space to do so, and the particles are not close enough to be attracted by the other particles.
Shape Break-Down: Since the particles of a solid are packed together, and constantly attracting to one another, these particles cannot move, and thus only stay in one shape (unless of course force is applied, or if it is heated or cooled). The particles of a liquid and gas do not have strong enough attraction, and thus the particles can re-arrange themselves, to take a new shape (not necessarily in a different amount of space, but in a different shape/form).
Volume Break-Down: Since all particles of matter always attract to one another; if the particles of a solid or liquid are constantly close enough to always have the same attraction to another specific particle, then that means that the particles will always be as close together to one another at all times (not necessarily in the same shape), and will always take up the same amount of space, at all times. So, a solid and a liquid, will always have the same volume, at all times. But for a gas, the particles aren't close enough to always have the same attraction to a specific particle, so the particles will not always be as close to one another. So, sometimes the particles will be close together, and take up less space, and sometimes the particles will be far apart, and take more space.
Ability to Flow Break-Down: Since the attraction between particles in a solid is quite strong, this prevents the particles from moving freely, so a solid's particles cannot flow. But, the bonds between particles in a liquid and gas, aren't as strong, so the particles can move freely, and take a new shape (with the same volume of course).
Changes in the States of Matter
Matter can change from a solid, liquid or gas, when it is heated or cooled. Heat causes particles to spread out, and move faster. So: solid--->liquid--->gas. And cooling something makes the particles come together, and move slower.
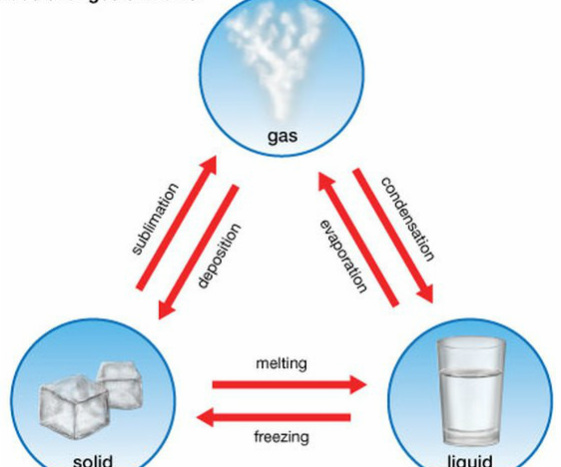
So: gas--->liquid--->solid. When matter changes, the particles stay the same, and the number of particles stays the same, but arrangement and movement does not. Below is a picture of all the different processes that occur to change states of matter:
So: gas--->liquid--->solid. When matter changes, the particles stay the same, and the number of particles stays the same, but arrangement and movement does not. Below is a picture of all the different processes that occur to change states of matter:
Particle Theory
There are 5 laws to particle theory, which all particles regardless of state of matter, composition, etc. follow:
1. All matter is made up of tiny particles.
2. All particles have empty spaces between them
3. Although you cannot see it, all particles are in constant motion
4. Particles move faster and spread apart when they are heated
5. All particles attract to one another
1. All matter is made up of tiny particles.
2. All particles have empty spaces between them
3. Although you cannot see it, all particles are in constant motion
4. Particles move faster and spread apart when they are heated
5. All particles attract to one another