Pure Substances vs. Mixtures
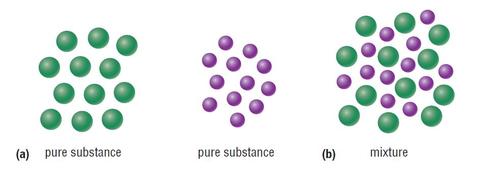
A pure substance is a substance comprised of only one type of particle. So, all the particles in the substance, are the same, and there is only one type. E.g. table salt is a pure substance as it is made up of only one type of particle, salt particle.
A mixture is anything that is made of two or more pure substances. You may think that anything that humans do not interfere with, is a pure substance. E.g. you may think cow milk is a pure substance, as that is the way it comes, and well, milk is only made of milk. But, if you thought that, you would be absolutely wrong. This is because even milk is made up of many different particles such as:
water
lactose
fat
protein
Mixtures can be found in all three states of matter: Steel (solid), tap water (liquid) and the air you breathe (gas). Below you can see a diagram of two pure substances being bonded to form a mixture (homogeneous mixture to be exact):
A mixture is anything that is made of two or more pure substances. You may think that anything that humans do not interfere with, is a pure substance. E.g. you may think cow milk is a pure substance, as that is the way it comes, and well, milk is only made of milk. But, if you thought that, you would be absolutely wrong. This is because even milk is made up of many different particles such as:
water
lactose
fat
protein
Mixtures can be found in all three states of matter: Steel (solid), tap water (liquid) and the air you breathe (gas). Below you can see a diagram of two pure substances being bonded to form a mixture (homogeneous mixture to be exact):
Compounds and Elements
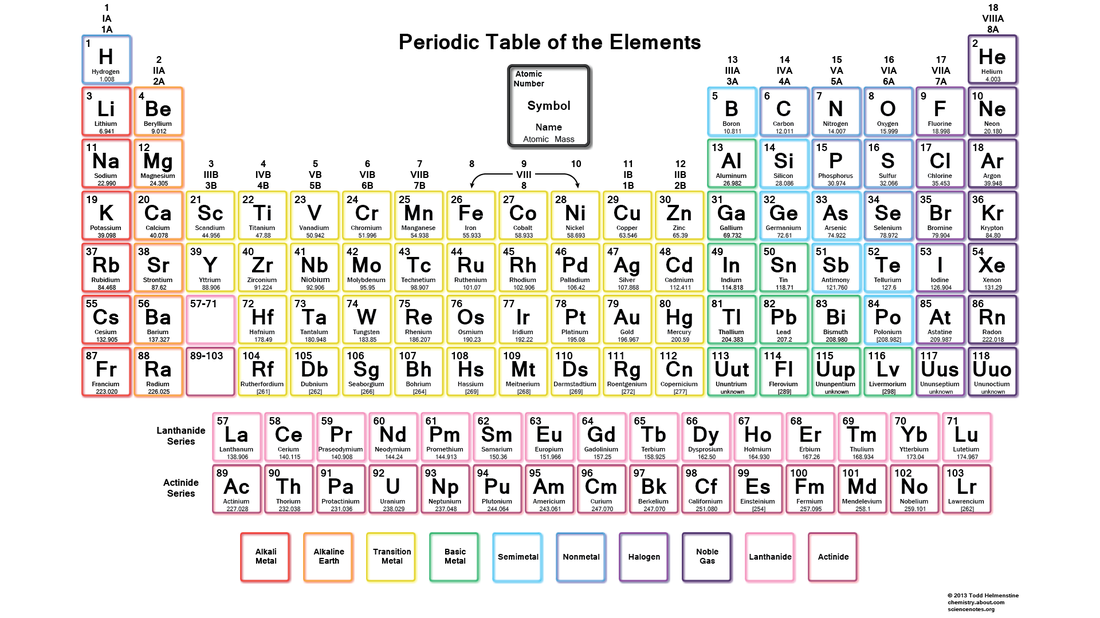
Elements: An element is something which is comprised of only one type of atom. Thus, an element cannot be further broken down. There are 118 elements currently known to us, which have all been listed below in the periodic table of elements. In the center of each box, is the symbol for that element, which is a short form for the name used internationally. Almost always only the first letter is capitalized, and usually comes form the Latin word for the element. Under the symbol is the name for the element, recognized and used everywhere in the world to avoid confusion. Under that is the atomic mass of the element, which is the weight of the protons, neutrons and electrons in a single atom of that element. The color of the box is explained at the very bottom of the chart/table.
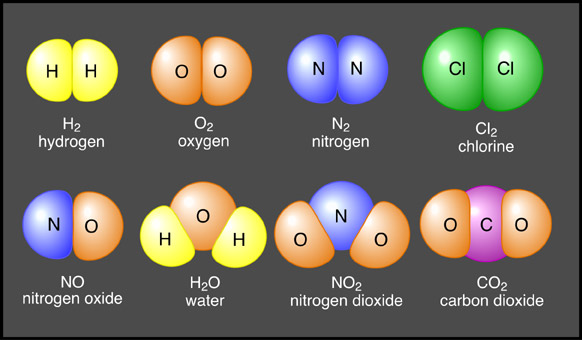
Compounds: A compound is something which is comprised of two or more atoms chemically bonded together. Because of this, a compound can be further broken down, although only chemically and not physically. Any specific compound will always have the same atom;atom ratio, unless it has been chemically tampered with. Here is an example of some compounds and their chemical compositions.
Mechanical Mixtures vs. Solutions
Both mechanical mixtures and solutions are two different types of mixtures. They both have different particles that make it up; but then what's the difference?
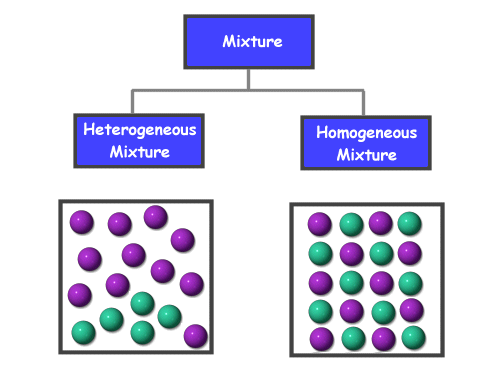
Mechanical Mixtures: Mechanical mixtures, also known as heterogeneous mixtures, are mixtures in which the particles are not chemically combined. Different substances are simply mixed with one another. It is often easy to tell the different substances apart in a mechanical mixture. E.g. the soil we find on the ground is a great example of a mechanical mixture. Soil is made up of sand, gravel, twigs, leaves, etc. which are simply put together, but not chemically bonded and thus easy to tell apart from one another. Another example would be sugar and salt. Even though if they were mixed together, it would be hard to tell the salt apart from the sugar (and vice versa) it would still be a mechanical mixtures as the substances have simply been mixed, not bonded.
Solutions: Solutions, also known as homogeneous mixtures, are mixtures in which the substance looks like a pure substance, but it is made up of many pure substances mixed together to look like one. You cannot tell that it is made up of many particles. E.g. we may think that juice or shampoo is a pure substance, as it is just juice or shampoo as you know it. But, both of these things are made up pure substances, and are a mixture, but look like a pure substance as the particles are in uniform. The very air you are breathing right now is also a solution. It is made up of pure substances such as nitrogen, oxygen etc., all mixed into one, and you cannot tell them apart from one another without appropriate equipment.
The reason of this difference is due to the fact that the particles in a mechanical mixture just layer out, and stay in groups with particles the same as it, so with the big groups it is easy to identify what it is. On the contrary, in solutions, the particles evenly mix and since you cannot see individual particles, this makes it all look like one. Below is a diagram which shows so quite well:
Mechanical Mixtures: Mechanical mixtures, also known as heterogeneous mixtures, are mixtures in which the particles are not chemically combined. Different substances are simply mixed with one another. It is often easy to tell the different substances apart in a mechanical mixture. E.g. the soil we find on the ground is a great example of a mechanical mixture. Soil is made up of sand, gravel, twigs, leaves, etc. which are simply put together, but not chemically bonded and thus easy to tell apart from one another. Another example would be sugar and salt. Even though if they were mixed together, it would be hard to tell the salt apart from the sugar (and vice versa) it would still be a mechanical mixtures as the substances have simply been mixed, not bonded.
Solutions: Solutions, also known as homogeneous mixtures, are mixtures in which the substance looks like a pure substance, but it is made up of many pure substances mixed together to look like one. You cannot tell that it is made up of many particles. E.g. we may think that juice or shampoo is a pure substance, as it is just juice or shampoo as you know it. But, both of these things are made up pure substances, and are a mixture, but look like a pure substance as the particles are in uniform. The very air you are breathing right now is also a solution. It is made up of pure substances such as nitrogen, oxygen etc., all mixed into one, and you cannot tell them apart from one another without appropriate equipment.
The reason of this difference is due to the fact that the particles in a mechanical mixture just layer out, and stay in groups with particles the same as it, so with the big groups it is easy to identify what it is. On the contrary, in solutions, the particles evenly mix and since you cannot see individual particles, this makes it all look like one. Below is a diagram which shows so quite well:
What is a Solution?
Solutions are everywhere! A solution is a when a type of particle dissolves into another. A mixture is when the two particles stay whole. E.g. iced tea may seem like a pure substance to you, but it is a solution as there are two particles, water and powder. One which dissolves (solute) and the other which the first dissolves into (solvent). Since there is less of the powder, and it is dissolving into the water, it is called a solute. Since there is more of the water, and it is being dissolved into, it is called a solvent, Here are the definitions of those big words:
Solute /'sälˌyo͞ot/
Noun
the minor component in a solution, dissolved in the solvent
Solvent /ˈsälvənt/
Noun
the liquid in which a solute is dissolved to form a solution
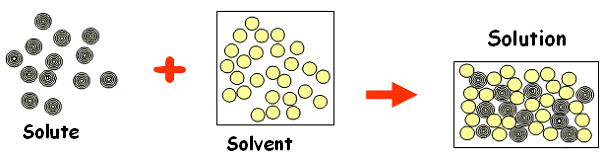
A solution is what you get when you dissolve a solute into a solvent. In a solution, both the particles of the solute and solvent are extant, but most often there is more of the solvent (more particles of the solvent than the solute), and so it is more visible/standing out. Common examples of solutions include brass and bronze. Brass is made when zinc is the solute, copper is the solvent, then brass is the solution. For bronze, tin is the solute, copper is the solvent, and bronze is the solution. Below is a picture of how solutes, solvents and solutions work:
Solute /'sälˌyo͞ot/
Noun
the minor component in a solution, dissolved in the solvent
Solvent /ˈsälvənt/
Noun
the liquid in which a solute is dissolved to form a solution
A solution is what you get when you dissolve a solute into a solvent. In a solution, both the particles of the solute and solvent are extant, but most often there is more of the solvent (more particles of the solvent than the solute), and so it is more visible/standing out. Common examples of solutions include brass and bronze. Brass is made when zinc is the solute, copper is the solvent, then brass is the solution. For bronze, tin is the solute, copper is the solvent, and bronze is the solution. Below is a picture of how solutes, solvents and solutions work:
As we all know, all particles have small spaces in between one another, corresponding to the size of the particle itself. So, when a group of big particles and small particles dissolve, the smaller particles fill in the space between the bigger ones. The reason these smaller particles get pulled into the bigger ones, is due to the fact that all particles attract to one another. So, that space which would have gone to waste, was used up by the smaller particles. So, the volume of a solution, is less than that of the solute and solvent summed up, due to the fact that the empty spaces which would otherwise go to waste, are being filled up.